On-Demand webinar with expert cardiologist Dr. Robert Shiroff
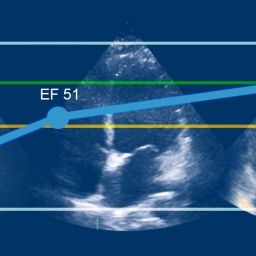
Dr. Robert Shiroff discusses the benefits of serial echos, when to do them, and how they can improve organ utilization. In some cases, when a heart has received an insult as a result of brain death, trauma or other factors, a series of echos (serial echos) can help determine if a heart is recovering, or if the damage is too much to transplant. Drawing from multiple case studies, Dr. Shiroff will show how tracking heart progress over time with a series of echos can be beneficial during donor management in determining a viable heart for transplant.
Who is This For?
Donor & Transplant Professionals
Robert "Bob" Shiroff, MD, FACC
CompuMed Chief Medical Director
Clinical Cardiologist
Dr. Shiroff is a board certified cardiologist with a fellowship in clinical pharmacology as well as an experienced medical expert witness.
CompuMed is honored to have Dr. Shiroff on our team as he serves the donor and transplant community. His in-depth knowledge and expertise helps assess, manage, and improve the recovery of donor hearts, resulting in an increase in the number transplanted into needy recipients. Knowing that each recovered heart has the opportunity to save a life is humbling and extremely rewarding. We are grateful for his passion to share his knowledge and experience with the OPO and Transplant community.